 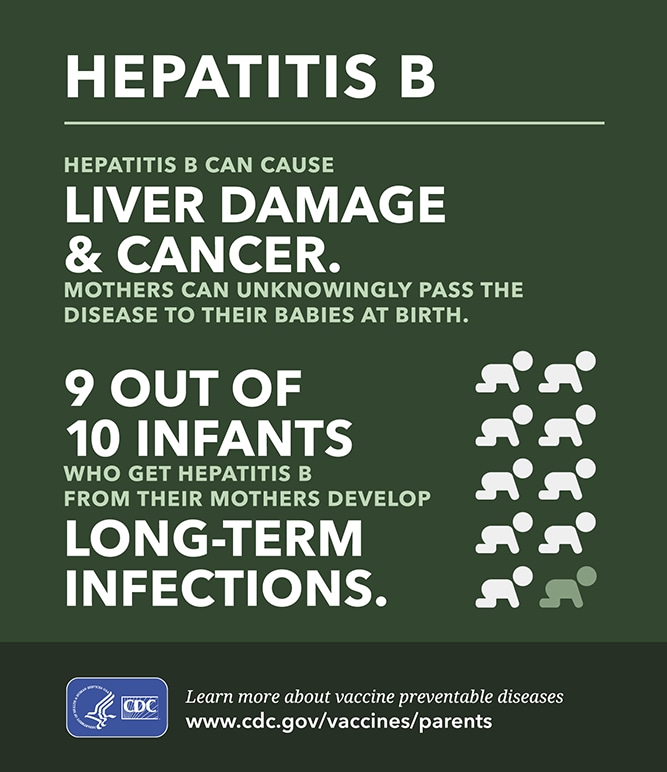
In countries with a high prevalence (>8 %) of HBsAg-positive persons, and before the implementation of universal immunization programs, most HBV infections were the result of early childhood household transmission or maternal-to-child transmission (MTCT). HBV is most infectious in persons with high-level viremia (HBV load) or positive HBeAg, a marker of high-level viral replication. Trial registrationĬ Identifier NCT01745822.Ĭhronic hepatitis B virus (HBV) infection affects an estimated 240 million persons worldwide, is complicated by cirrhosis of the liver and hepatocellular carcinoma, and causes 650,000 deaths annually. The results of this randomized trial will clarify the efficacy and safety of a short course of antiviral treatment to prevent mother-to-child transmission of HBV and inform international guidelines. Secondary endpoints are maternal and infant adverse events, acute exacerbations of maternal hepatitis B disease (ALT >300 IU/L, defined as a “flare”) following discontinuation of study treatment, infant HBV infection status and growth up to 12 months of age. The primary infant endpoint is detection of HBsAg, confirmed by detection of HBV DNA at six months of age. 
Mothers and infants are followed until 12 months after delivery. The target sample size of 328 mother/infant pairs assumed 156 evaluable cases per arm to detect a ≥9 % difference in MTCT transmission (3 % experimental arm versus 12 % placebo arm) with 90 % power. Participant women at study entry must be age ≥18 years, hepatitis B surface antigen (HBsAg) and e-antigen (HBeAg) positive, have alanine aminotransferase (ALT) level 50 mL/min, and no history of anti-HBV antiviral treatment. All infants receive HBIg at birth, and a hepatitis B (HB) vaccination series according to Thai guidelines: birth, and age 1, 2, 4 and 6 months. The study is a phase III, multicenter (17 sites in Thailand), placebo-controlled, double-blind, randomized 1:1, two-arm clinical trial of TDF 300 mg once daily versus placebo among pregnant women from 28 weeks’ gestation through 2-month post-partum. 
We designed a study to assess the efficacy and safety of a short course of maternal tenofovir disoproxil fumarate (TDF) among women with a marker of high viral load for the prevention of MTCT of HBV. The risk of mother-to-child transmission (MTCT) of HBV has been estimated to be about 12 % when mothers have a high hepatitis B viral load, even if infants receive passive-active prophylaxis with HBV immunoglobulin (HBIg) and initiate the hepatitis B vaccine series at birth. In Thailand, 6-7 % of adults are chronically infected with HBV.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
